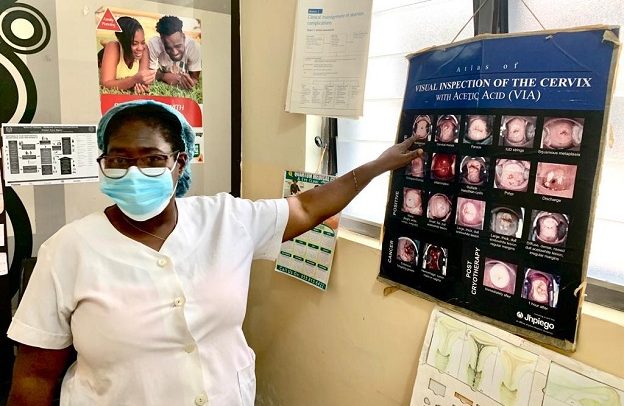
Madam Abigail Ogunro-Yorke pointing to the visual inspection of the cervix chart
“You see this whitish part or lesion in the cervix, it tells you that there is a presence of the Human Papilloma Virus (HPV)” says Madam Abigail Ogunro-Yorke, senior midwife and head of facility at the Planned Parenthood Association Ghana (PPAG) Family Health Clinic, located in Lartebiokoshie, a suburb of Accra.
Madam Ogunro-Yorke says patients who have precancerous lesions are at the early stages of detection and are advised to undergo treatment as soon as possible, ‘if not, in the next five to 10 years, it will become cancerous and little can be done.’
The senior midwife who attends to a number of girls and women with varied sexual and reproductive health needs says her advice to young girls who are sexually active and women who test negative for HPV is to get the HPV vaccination as soon as possible to protect themselves.
She, however, notes that the clinic does not offer HPV vaccination services and that patients who need to get vaccinated are directed to the Korle Bu Teaching Hospital (KBTH) for the service.
The HPV vaccine for adolescents was more widely introduced in Ghana in 2013 with support from a campaign through Gavi, the Vaccine Alliance and the Danish government.
The national vaccination campaign to protect young girls against cervical cancer under the initiative of former First Lady, Lordina Mahama, saw about 33,000 school girls vaccinated in the pilot programme.
However, the challenge of cost of HPV vaccination has made many young girls and women delay vaccination thereby leaving them exposed to the HPV infection.
Pull Out
Cervical cancer is a type of cancer that occurs in the cells of the cervix – the lower part of the uterus that connects to the vagina. It is mostly caused by the sexually transmitted human papilloma virus (HPV).
According to the Catalan Institute of Oncology (ICO) and the International Agency for Research on Cancer (IARC) Ghana has a population of 10.6 million women aged 15 years and older who are at risk of developing cervical cancer.
Current estimates indicate that every year, 2,797 women are diagnosed with cervical cancer and 1,699 die from the disease.
Cervical cancer ranks as the 2ndmost frequent cancer among women in Ghana and the 2nd most frequent cancer among women between 15 and 44 years of age.
Spokesperson for Gavi, the Vaccine Alliance, says the HPV vaccine is one of the most impactful vaccines, with 17.4 deaths averted per 1000 young adolescents vaccinated.
However, limited supply of the HPV vaccine has been a challenge over the last few years due to the small number of HPV vaccines prequalified by the World Health Organisation (WHO), and not enough doses being produced to meet global demand.
Single Dose HPV Vaccine
In April 2022, following a recommendation of the WHO Strategic Advisory Group of Experts on Immunization (SAGE), the WHO released a new position paper indicating that a single-dose schedule, referred to as an alternative, off-label single–dose schedule can provide a comparable efficacy and durability of protection to a two-dose regimen.
The optimization of the HPV schedule is expected to improve access to the vaccine, offering countries the opportunity to expand the number of girls who can be vaccinated and alleviating the burden of the often complicated and costly follow-up required to complete the vaccination series.
The Gavi spokesperson says the WHO recommendation is good news as it potentially ‘frees up’ more doses to reach more girls, presenting a unique opportunity to accelerate progress toward reaching vaccination targets.
“The good news is that due to increased production and new vaccines coming to the market, the outlook is favourable, and supply is expected to triple in the next few years,” the spokesperson adds.
Clinical Trial Evidence
Results are emerging from several clinical trials evaluating the ability of a single dose HPV vaccine to elicit a strong immune response against incident and persistent HPV infections, which are the necessary prerequisites to development of cervical lesions and in the longer term, cervical cancer.
Single Does HPV Vaccine Evaluation Consortium’s publication of the evidence and potential impact for single-dose HPV vaccination shows that single-dose HPV vaccination could substantially reduce the incidence of HPV attributable cervical precancer and cancer.
Also, data accumulated from clinical trials and high-quality observational clinical studies shows comparable efficacy and effectiveness between single and multidose schedules in preventing HPV infections, lasting up to 10 years following vaccination.
For instance, a randomized controlled trial in Kenya with girls and women aged 15-20 showed single-dose vaccination with Gardasil®9 or CervarixTM was about 98% effective in preventing HPV 16/18 persistent infections.
The data also shows that the immune response 24 months post-vaccination of a single dose in girls aged 9-14 in Tanzania was non-inferior to a single dose in historical cohorts for which single-dose efficacy was shown.
Thus, compared no vaccination, single-dose HPV vaccination yields substantial health benefits and is good value for money.
“Reaching more girls with a single dose will avert much more cervical cancer cases than vaccinating fewer girls with a second dose. Immediate implementation of a single-dose HPV program leads to greater health benefits than delaying implementation,” the Consortium asserts.
“Increasing the availability of affordable HPV vaccines and expanding options globally is crucial for reaching WHO coverage goals,” says Anne Schuind, MD, PATH Initiative Team Leader for HPV Vaccines. “HPV vaccination is highly effective, and all girls deserve access to this lifesaving tool—no matter where they live.”
She says PATH is conducting a series of country-specific cost-effectiveness analyses that can help local leaders estimate long-term health, economic, and financial impacts and make decisions about which vaccine products will be most appropriate and sustainable.
“WHO’s goal is both worthy and attainable. Achieving it will mean saving hundreds of thousands of women’s lives from cervical cancer, but only if we vaccinate as many girls as possible today—and we at PATH are committed to doing our part,” she adds.
Ghana HPV Vaccine Trial
Professor Tsiri Agbenyega, is a lecturer at the Kwame Nkrumah University of Science and Technology (KNUST) and a Principal Investigator (PI) at the Malaria Research Centre, Agogo Presbyterian Hospital.
He and his team are currently undertaking a phase three trial on a vaccine against the Human Papilloma Virus (HPV).
Infection with HPV has been shown to cause cervical cancer, which is one of the most common cancers among women in Ghana.
Cecolin, the HPV vaccine they are studying, is not licensed in Ghana yet, but it has been prequalified (approved) by the World Health Organization in 2021.
The trial, sponsored by the global health organization PATH, is being undertaken among young girls aged 9 to 14 years, and is being done in order to show that the vaccine works similarly in different countries and using different schedules.
The study would therefore provide public health officials with evidence regarding an additional safe and effective option for HPV vaccination that is expected to be available at a lower cost to governments.
He says that, as required for all clinical trials; the research team submitted the trial documents, including consent process, to the Food and Drugs Authority (FDA), the Ghana Health Service (GHS) Ethics Committee as well as the Ethics Committee of KNUST for approval before commencing the trial.
All girls had to have consent from their parents or guardians in order to participate.
“The trial is about two thirds done and the findings will be shared soon after the end of the trial which is anticipated in late 2024,” Prof. Agbenyega noted.
Funding Support
Gavi spokesperson says understanding the cost-effectiveness of different HPV vaccine options is essential for countries considering the introduction of an HPV vaccine or switching to a different product.
“Countries’ regulators will need to evaluate the data and decide if they prefer to go with an ‘off-label’ 1-dose schedule or 2-dose schedule.
Once they have made that decision, countries eligible for Gavi support to introduce the HPV vaccine can do so on either dose schedules. Countries that have already introduced the vaccine on a 2-dose schedule and would like to switch to a 1- dose schedule are eligible for a switch grant from Gavi,” the spokesperson assures.
(This reporting was supported by the International Women’s Media Foundation’s Global Health Reporting Initiative: Vaccines and Immunization in Africa, in partnership with Sabin Vaccine Institute.)
By Jamila Akweley Okertchiri